REPACKAGED BEVACIZUMAB
Solutions designed with your practice in mind
As the #1 503B provider of repackaged bevacizumab, Pine has over a decade of experience providing reliable repackaged bevacizumab* services to practices nationwide.
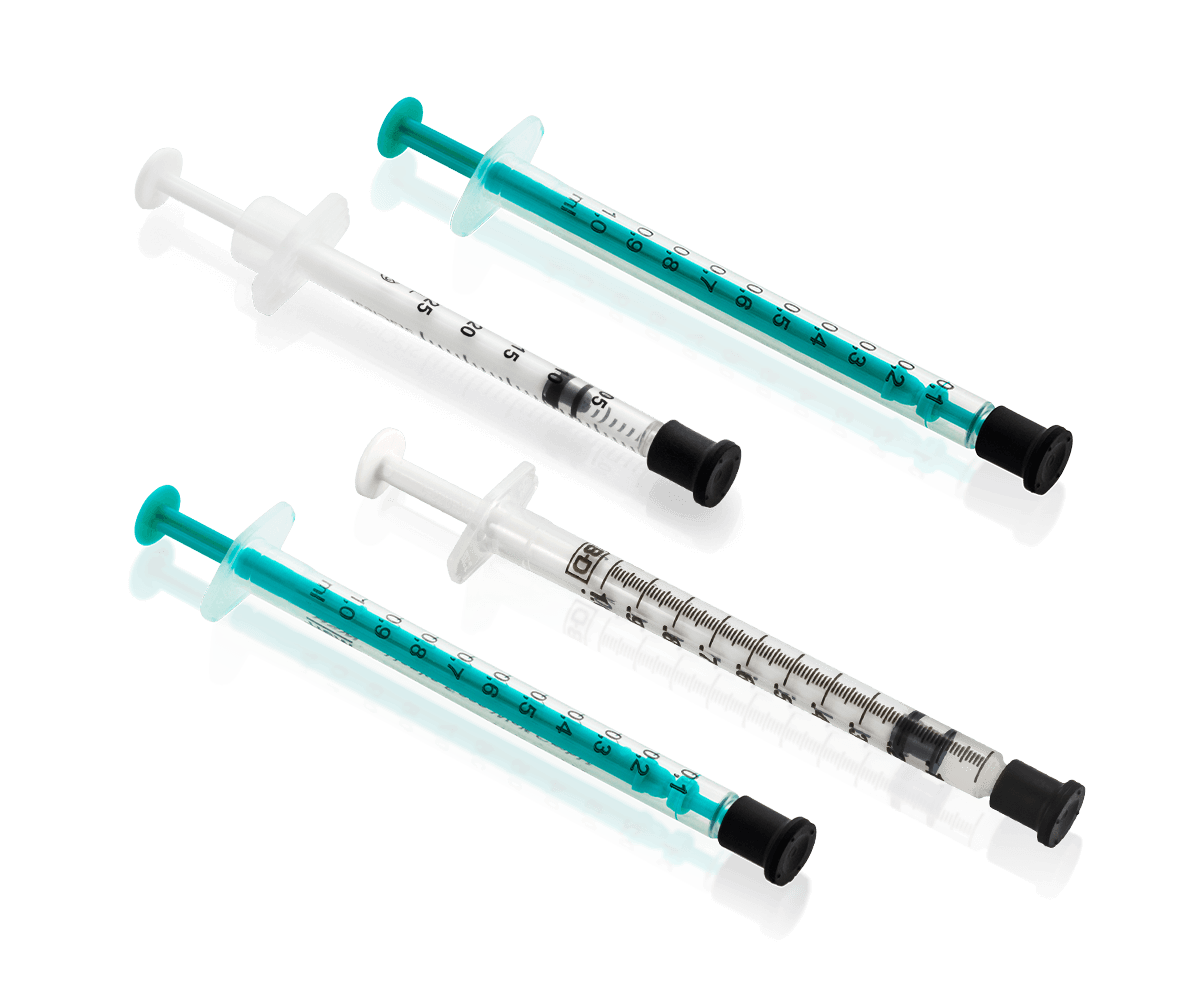
As our partner, you always have options
Pine provides a wide selection of syringe options for repackaged bevacizumab*, giving your facility the flexibility to provide the quality care your patients deserve.

*Bevacizumab is not FDA-approved for intravitreal use. These statements have not been evaluated by the Food and Drug Administration. This product is not intended to diagnose, treat, cure, or prevent any disease.
